 
Most people can safely get the Pfizer/BioNTech COVID-19 vaccine.īut this vaccine is not recommended if you: Pfizer/BioNTech - extremely rare side effects Signs of myocarditis in younger children may be more general including: palpitations (a forceful heartbeat that may be irregular).Get urgent medical help if you get any of these symptoms in the weeks after the Pfizer/BioNTech vaccine: We do not know the risk of myocarditis or other rare side effects after a booster dose yet. 1 additional case for every 17,500 men aged 16 to 24 (within 28 days).1 additional case for every 38,000 men aged 12 to 29 (within 7 days).The risk of these very rare conditions is higher in young men.Ģ European studies have estimated the risk of myocarditis in men, after the second dose of the vaccine as: They have mostly been seen within 14 days of getting the vaccine. These conditions are more likely to happen after the second dose. Myocarditis and pericarditis are inflammatory heart conditions. Very rare side effects may affect up to 1 in 10,000 people. The Pfizer/BioNTech COVID-19 vaccine is called Comirnaty. 
Pfizer/BioNTech COVID-19 vaccine side effects The side effects of the COVID-19 vaccines used in Ireland are listed below. If you have had anaphylaxis (severe allergic reaction) to Trometamol, you should not get the adapted mRNA vaccines. The safety of the vaccines will continue to be monitored by the European Medicines Agency (EMA). We expect that the side effects of the adapted (bivalent) mRNA vaccines will be similar to the previous Pfizer/BioNTech and Moderna COVID-19 vaccines. 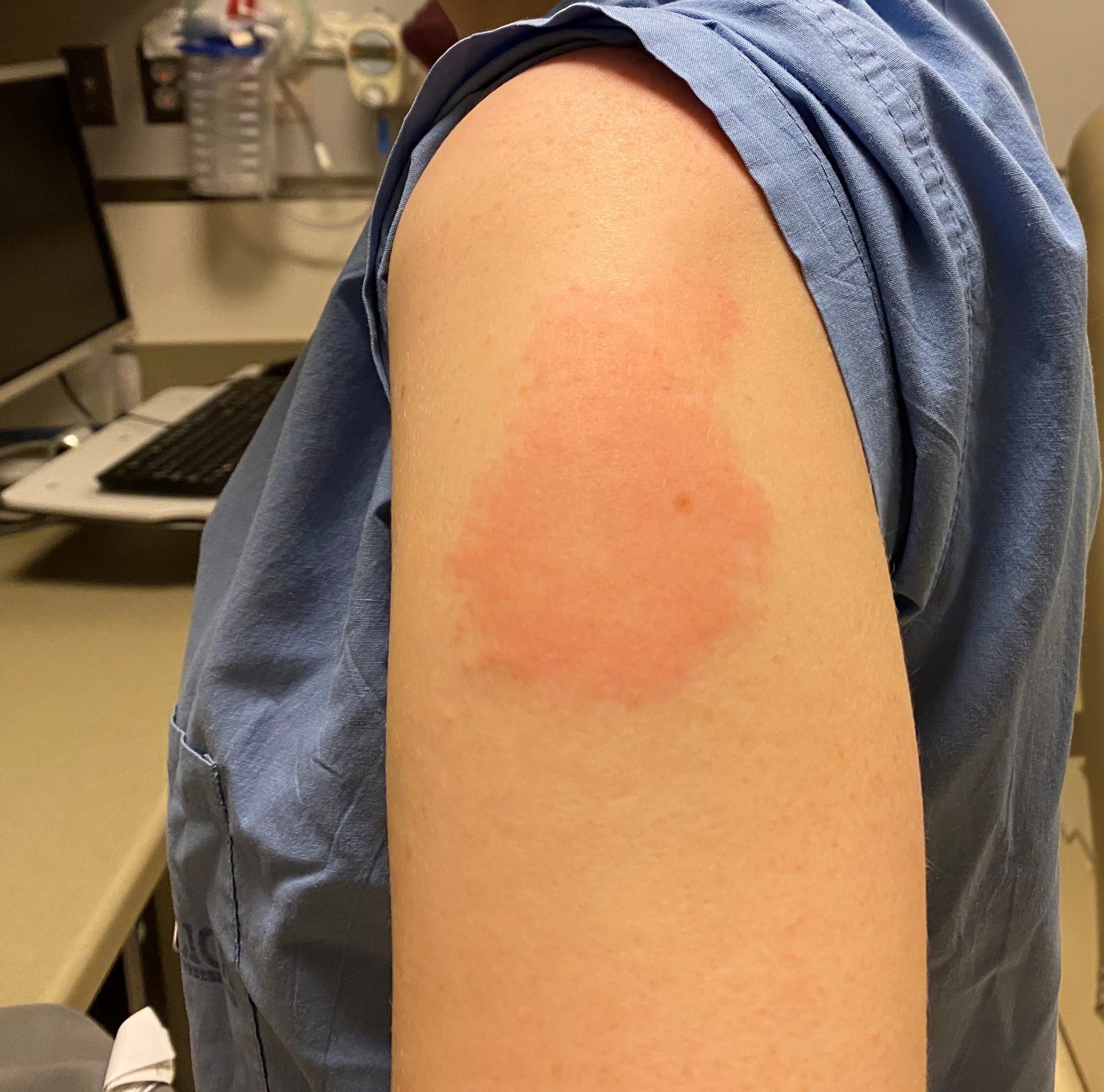
There have not been any unexpected side effects for people who have had multiple boosters. Many countries have given multiple doses of COVID-19 vaccines. If you feel uncomfortable, take paracetamol or ibuprofen following the instructions on the box or leaflet. This usually happens within 48 hours of getting a vaccine. It’s common to develop a fever (temperature of 38 degrees Celsius or above) after COVID-19 vaccination. Your vaccinator is trained to treat allergic reactions. Serious side effects, like a severe allergic reaction, are extremely rare. If you are concerned about side effects, phone your GP for advice. Most of these are mild to moderate and short-term. These were predominantly mild or moderate in intensity and resolved on their own within a few days, according to clinical trial results presented by Moderna.Like all medicines, COVID-19 vaccines can cause side effects. The company said this smaller dose still produced good immune responses and would increase how many doses Moderna can produce.Įven with a smaller dose, people who received the booster shot still experienced common side effects. The initial two injections were 100 micrograms the third shot would be 50 micrograms. Moderna plans to use a half-strength dose for its third shot. The FDA typically follows the advisory group's recommendations. The agency's expert panel unanimously recommended Thursday OK'ing a third jab for certain high-risk groups, including people 65 years and older as well as younger people at higher risk of severe disease. The Food and Drug Administration is in the final stages of reviewing Moderna's booster-shot application. It often indicates a user profile.Ī booster shot of Moderna's coronavirus vaccine has similar side effects to what people experienced after the second dose. Account icon An icon in the shape of a person's head and shoulders.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
